thought leadership
What are cell and gene therapies and what are the key considerations for market access?
June 5, 2023
In the Asia-Pacific (APAC) region, the demand for cell and gene therapies (CGTs) is increasing but the reimbursement environment for these therapies is still nascent. This update, taken from our expert insights report, introduces CGTs and shares 4 reasons why access for CGTs is different from traditional therapies.
What are cell and gene therapies?
CGTs are therapies that involve the modification of genetic information in living cells. They are often intended to treat, modify, reverse, or cure a life-threatening disease.
The terms cell therapies and gene therapies are often used interchangeably and the definitions can become blurred; this report adopts the definitions illustrated in the following graphic.
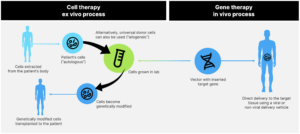
Cell therapy refers to the genetic modification of cells ex vivo—that is, outside of the body. Cells are extracted from a patient, gene transfer is performed in a lab, and the cells are then reintroduced back into the body. This is known as autologous cell therapy. More recent ex vivo approaches have involved allogeneic cells, which are genetically modified donated cells that are cultured and transplanted into the patient.
Gene therapy involves the in vivo delivery of genes directly to cells inside the body.
CGTs can also include minimally manipulated and homologous products (such as tissue cultures, xenografts, etc.), but this update will focus on more than minimally manipulated and non-homologous “curative” therapies.
4 reasons why market access for cell and gene therapies is different from traditional therapies
While CGTs offer hope for patients with complex diseases, enabling access for CGTs poses unique challenges that are different to those for traditional therapies.
1. Complex manufacturing flow, compounded with logistical constraints, limit CGT availability
Manufacturing of CGTs is a complex and intricate process, requiring harvesting cells, shipping patient samples to and from the manufacturing center, all while ensuring product quality. The highly resource-intensive steps involved are a hurdle to ensuring their widespread availability.
2. Sophisticated treatment delivery involving continuous patient monitoring with multiple drop-off points
Unlike traditional therapies, which patients can often collect from a pharmacy, CGTs may have multiple drop-off points due to subjective standards for determining eligibility for a CGT or a lack of access to accredited facilities eligible to provide the therapy, as well as limited capacity for follow-on monitoring and care.
This requires sophisticated healthcare delivery systems to tackle awareness gaps and ensure seamless patient diagnosis, selection, monitoring, and post-treatment care, along with appropriate training of healthcare personnel and cold-chain infrastructure to meet required standards.
3. Ultra-high cost paid over short duration, with multiple payment touch points pre- and post-infusion
Another distinctive aspect of CGTs is their ultra-high costs. CGTs are “one-off” therapies intended to cure diseases in a single treatment, with the aim of eliminating (if successful) the persistent costs associated with chronic conditions. However, the financial burden to patients can be substantial, ranging from US$500,000 for chimeric antigen receptor T-cell (CAR-T) therapies to over US$1 million for adeno-associated virus (AAV)-based gene therapies, with costs often incurred over a short duration.
Additionally, the costs outside of the actual CGT infusion, involving the diagnostics, process of determining CGT eligibility, and post-infusion care, necessitate multiple payment touch points. Thus, the immediate financial strain on patients—especially if they bear CGT costs primarily out-of-pocket—can be substantial.
4. Uncertainty with regards to long-term treatment outcomes
Lastly, unlike traditional therapies with well-established track records, the relatively novel nature of CGTs introduces uncertainty, with long-term outcomes needing to be monitored closely. The recent class-wide CAR-T boxed safety warning mandated by the US Food and Drug Administration (FDA), is a testament to the fact that these lifechanging treatments are still new, warranting transparent and prompt information exchange. Over the 7 years since the launch of the first CAR-T therapy, the uncertainty around treatment outcomes has impacted decision-making by healthcare providers, regulators, payers, and the other stakeholders involved.
Navigating the complexities of market access for cell and gene therapies required innovative solutions
CGTs are a distinct set of therapies, in fact many global bodies have even classified CGTs as advanced therapies that may need to be considered more akin to procedures, involving some elements of what have been traditionally seen as drugs and devices, combined. Navigating the complexities of manufacturing, patient-tracking, cost structures, and treatment outcomes requires tailored logistical and financing support through innovative solutions to cover the full breadth of challenges that CGTs face.
