thought leadership
What is market access?
August 20, 2023
For biopharmaceutical and medical technology manufacturers, enabling faster patient access to therapies is a key challenge. An effective market access strategy helps them solve the challenge.
Market access: a basic definition
In the simplest terms, market access is the process to ensure that all appropriate patients, who would benefit from a new therapy, get rapid and maintained access to the right treatment, at a price that reflects its value.
It’s about:
- Defining value for the patient population
- Communicating data in support of the therapy to prove additional value to the patient and the healthcare system.
Within the biopharmaceutical and medical technology industries, this relates to prescription medicines including vaccines, diagnostics, medical devices, and digital therapeutics.
MALCOM eLearning platform
This film, and more like it, is available to watch within MALCOM, the eLearning platform for HEOR and market access professionals.
Why is market access important?
Providing healthcare has become a costly business and the challenge to healthcare systems to fund treatments and healthcare is enormous.
Healthcare systems simply cannot provide every therapy available to them. As a result, payers, who are the gateway to funding within the healthcare systems, allocate the budget available to deliver a healthcare service, and the treatments available therein.
Therefore, biopharmaceutical and medical technology manufacturers effective market access strategies that communicate value and support reimbursement so that patients have access to new, effective therapies as soon as possible.
What does successful market access look like?
In the biopharmaceutical and medical technology industries successful market access can be explained as the achievement and retention of an appropriate value position in a healthcare system’s list of recommended treatments and medical technologies. Put simply, the intervention is reimbursed as a treatment of choice.
The payer requirements and processes involved in order to make decisions vary for different categories of interventions. However, the principle of market access—the discipline to plan for and implement activities that focus on the financial decision-making of payers—is the same.
Market access can be a complicated process.
Manufacturers need to communicate the value of their product to many stakeholders before their drug can be accessed by patients, and this process differs between countries.
This simplified model illustrates the important role of market access within the process.
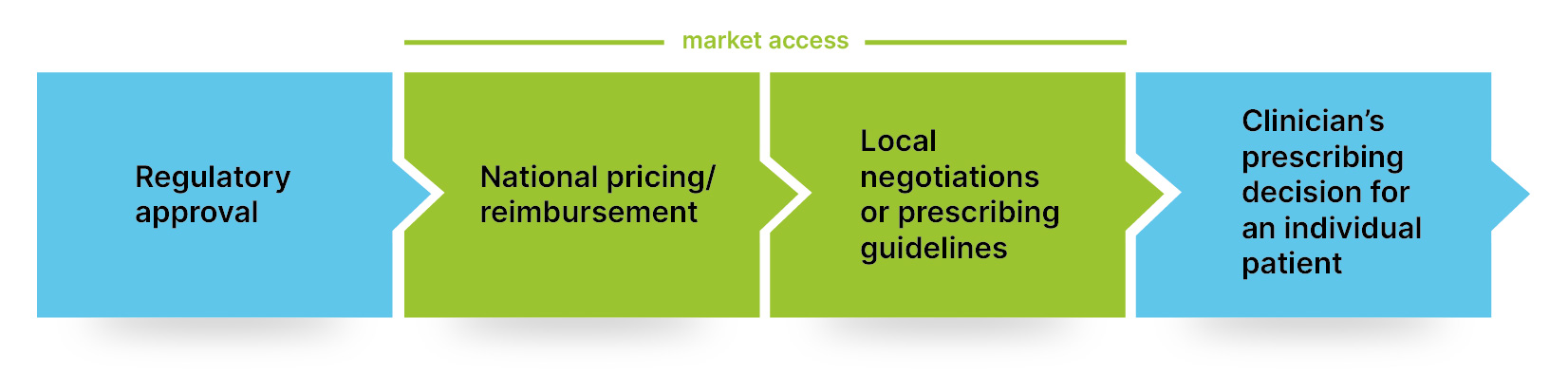
Communicating value to healthcare stakeholders
- Regulatory approval: The manufacturer obtains approval from regulators such as the European Medicines Agency (EMA) and the Food and Drug Administration (FDA). Approval permits the manufacturer to sell the drug.
- National pricing/reimbursement: Even if licensed, the drug may not be accessible to patients. In many countries the price and the target patient for the drug needs to be agreed before patients can access the drug.
- Local negotiations or prescribing guidelines: Locally or at a hospital level there can be variety in decision-making. For example, a country may have 30 licensed drugs available for a condition, which are all reimbursable. But if the local prescribing guidelines say the patient can’t have one of these drugs, they probably can’t.
- Clinician’s prescribing decision for an individual patient: Ultimately, the clinician decides which drugs they prescribe to a patient. That can be determined by many factors such as guidelines, local formularies, or how a previous patient responded to that drug.
This shows that there is more to patient access than getting a regulatory approval for a drug and then working with clinicians to prescribe it. The stages in between need the same consideration, and this is where an effective market access strategy is required.
How do pricing and reimbursement decisions support market access?
Pricing and reimbursement are closely linked with market access. They support the process by making sure that the drug can be prescribed to the appropriate patients and reimbursed by the healthcare system.

- Pricing: the list price, or national price, of the drug.
This is the published price that needs to be paid to the manufacturer. There are elements that can be added and taken out, such as charges from wholesalers or hospital-negotiated discounts and agreements.
Many countries set the price they are willing to pay by looking at the prices in other countries. For example, the prices in France, Germany, and the UK are commonly referenced and will influence the price paid in other countries. - Reimbursement: gaining payment for the drug. How the treatment is paid for differs between countries and, where applicable, different health technology assessment (HTA) systems. This is the subject of continual development and change.
Pricing and reimbursement is complex and subject to country-specific requirements. In some countries pricing and reimbursement go together, in some countries they are very separate.
Creating the connections between biopharmaceutical and medical technology manufacturers and healthcare stakeholders
Beyond the safety, efficacy, and quality of the licensed therapy, manufacturers need early understanding of the reimbursement and price potential for their product from public healthcare systems.
Healthcare systems are faced with increasingly difficult choices. The payers in these systems are driven to:
- Control new product endorsement and ensure the most efficient use of the drug
- Devise new methods to control costs such as managed entry agreements, evidence-based pricing, and volume limits
- Consider patient demand in terms of the need for effective medications, and in the future, willingness to pay.
As a result, biopharmaceutical and medical technology industries must ensure that the value of new drugs is clearly explained in terms that are relevant to country-specific healthcare payers and stakeholders.
Policy, access, value, and evidence partners, like the experts at Avalere Health, can help manufacturers determine, support, and communicate this value.
How does a policy, access, value, and evidence partner support manufacturers
Biopharmaceutical and medical technology manufacturers connect with commercialization partners, such as Avalere Health, to help them solve their market access challenges.
Services that we provide include:
Global market access
From the US to Asia-Pacific, and across Europe, clients depend on our best-in-class capabilities to optimize access and demonstrate the full value of their innovative products to payers. Our multidisciplinary experts and digital visionaries are shaping the market. Read more.
Global HEOR
From early-stage development through to successful launch and beyond, our HEOR capabilities provide robust analytics and evidence-based strategic insights to support the value of assets—helping inform what healthcare should be provided, how it should be delivered, and to whom. Read more.
Global value engagement
Bringing value to life through compelling, insight-driven, evidence-based strategy and communication that makes the complex simple, engages stakeholders, and ensures the value of your assets stands out from the crowd. Read more.
Federal and state policy
Our team of healthcare policy, regulatory, and legislative experts will guide you through the complexities of all aspects of federal and state healthcare policies and help you prepare for how changes will impact coverage, access, payment, care delivery, and consumer choice. Read more.
Due diligence
Thinking like investors, we deliver customized, deep, data-driven analysis to quantify relevant risks, opportunities, and trends to inform financial models and potential goal achievement. Read more.
The digital transformation of market access.
In recent years, we have seen how digital technology, artificial intelligence, and big data have been harnessed to improve health outcomes.
With the rapidly evolving market access landscape, we are now on the cusp of a huge acceleration in the use of digital applications to explore value options, gain strategic advantage, and provide smarter market access solutions.
Our suite of digital applications is transforming market access processes for leading biopharmaceutical and medical technology manufacturers.
Take a tour of our secure and intuitive cloud-solutions.
This learning and more are available within MALCOM, the eLearning platform for HEOR and market access professionals
MALCOM is an on-demand eLearning platform that helps market access teams of all sizes to upskill and keep their finger on the pulse of evolving value, evidence, and access landscapes.
