thought leadership
Navigating the evolving HTA landscape for medical devices in the Asia-Pacific region
Carol Kao, Senior Consultant; Smarth Lakhanpal, Associate Director | 4/16/2024
In the Asia-Pacific (APAC) region, health technology assessment (HTA) plays an increasingly important role in evaluating new medical devices. This update highlights four key success factors for optimizing access to innovative medical technologies and offers valuable insights into navigating the evolving HTA landscape in APAC.
The APAC region is home to 5 of the world’s 15 largest medical device markets and is the second largest region by total sales. High-income markets in the region (Japan, South Korea, Australia) spend as much as, or in some cases more than, Organisation for Economic Co-operation and Development (OECD) markets on health (an average of 10.2% of GDP is spent on healthcare by high- income APAC markets, which is greater than the OECD average of 9.7% in 2021).
Table 1: APAC key markets outlook
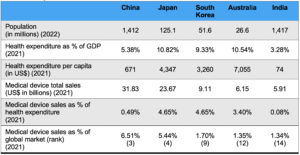
Source: Avalere Health analysis, WHO global health expenditure database, Fitch Solutions, Worldwide Medical Devices Market Factbook
Governments across the APAC region face a complex challenge: ensuring access to innovative medical technologies while maintaining an efficient and sustainable healthcare system. HTA is beginning to play a crucial role in navigating this challenge by providing a systematic evaluation framework for medical devices. With the increasing trend towards adoption of HTA, manufacturers are increasingly required to demonstrate value for money to achieve reimbursement. This article sheds light on the current state of HTA for medical devices in APAC, highlighting the critical success factors for optimizing access.
Diverse landscape in APAC: a patchwork of HTA systems
While the importance of HTA is widely recognized, there is no single, unified framework across the APAC region. Each country has developed its own approach, reflecting specific healthcare priorities and resource constraints. However, some countries stand out for their well-established and continuously evolving HTA systems for medical devices. Australia, South Korea, and Japan, with their large medical technology markets, serve as prime examples where the application of HTA for medical devices goes beyond simply assessing a product’s technical merits and includes other metrics such as health equity, patient perspective, and cost-effectiveness.
In contrast, some middle- and low-income countries often prioritize cost-minimization and budget impact, leading to limitations in the coverage of innovative medical devices. This can restrict patient access to the latest technology.
Figure 1: Different paths to balancing equitable access in APAC

Beyond national HTA
Not just at a national level but even at a local hospital procurement level, the need to demonstrate value for money has become quintessential to gaining in-market access. Value-based purchasing and procurement is becoming mainstream in markets such as Australia and Singapore. Criteria beyond cost of treatment alone to award contracts, for example the impact on patient outcomes, ease of healthcare delivery, and management of the total cost of care are becoming key considerations in procurement decisions.
Critical success factors for optimizing patient access to innovative medical devices in APAC
The HTA landscape in APAC presents both challenges and opportunities. By fostering a collaborative approach between governments, industry, and patient advocacy groups, the region can ensure that HTA effectively promotes access to innovative medical devices while optimizing healthcare resource allocation.
The critical success factors for shaping the assessment and optimizing access include:
- Establishing a common definition of value across stakeholders
- Going beyond the clinical and cost-effectiveness arguments and incorporating the broader societal impact of innovation to establish value
- A commitment to transparency and evidence-based decision-making
- Understanding patient needs: engaging with patient advocacy groups and understanding the perspectives of patients and caregivers to align with those who benefit most from new technologies.
Creating the connections that make better health happen
At Avalere Health, we partner with members of the APAC ecosystem to advocate for the pivotal role of industry in engaging with various stakeholders to develop methodologies that better recognize the value of innovative technologies.
In February 2024, the APAC team at Avalere Health shared their insights in a webinar on “HTA for Medical Devices in Asia-Pacific” hosted by APACMed, where best practices from established HTA markets to achieve access to innovative treatments were explored.
For a closer look at the key takeaways and the learnings for the industry, please visit the APACMed website or download the webinar summary.

