thought leadership
Commercializing and funding global cell and gene therapies in the Asia-Pacific region
Vesshnu Sutharsan, Consultant; Sudha Sundaram, Associate Director | January 1, 2024
In the Asia-Pacific (APAC) region, the demand for cell and gene therapies (CGTs) is increasing but the reimbursement environment for these therapies is still nascent. This update and excerpt from our on-demand webinar, summarizes the different approaches taken to commercialize and fund global CGTs in APAC.
Market entry models for cell and gene therapies in Asia-Pacific markets
The specialized nature of CGTs requires more reliance on the local healthcare ecosystem compared with traditional therapies. Global companies have hence had to explore market entry models beyond setting local subsidiaries, including licensing, strategic alliances, and joint ventures with existing local stakeholders.
For example, in China, Novartis entered into a strategic alliance for Kymriah where CBMG manufactures and supplies Kymriah in China and receives a mark-up on sales, while Novartis exclusively retains the license in China.
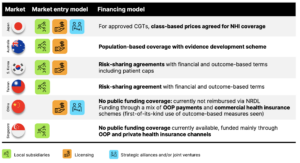
Funding decisions for CGTs remain largely budget-impact driven across APAC, with the exception of value assessments in Australia.
Japan: Managed entry agreements have not yet been implemented. Approved CGTs are automatically covered by national health insurance but at a class-based price, with co-payments accounting for 30% of the total medical cost for patients under 70 years of age.
Australia: A population-based coverage with an evidence development scheme has been implemented for chimeric antigen receptor T-cells (CAR-Ts), which have been assessed for funding as a procedure rather than as a therapy. This specialized arrangement for “Highly Specialized Therapies” is jointly funded by national, state, and territory governments.
South Korea and Taiwan: Kymriah and Zolgensma are subject to price-volume and performance-based agreements with annual per-patient expenditure caps in South Korea. It has also been reported that additional confidential rebates on list price were negotiated for these therapies, referencing corresponding prices in Japan. Taiwan has recently announced coverage for the same therapies employing financial and outcomes-based risk-sharing agreements as well.
Singapore and China: There is currently no public funding coverage for CGTs; these therapies are funded primarily through out-of-pocket payments and private or commercial health insurance channels. Notably, while China has covered innovative therapies on its national reimbursement list based on substantial discounts in the past, no CGTs have qualified for inclusion in the National Reimbursement Drug List (NRDL) because manufacturers have not met the required discount ranges.
Nonetheless, a first-of-its-kind innovative outcome-based insurance scheme has been announced for Yescarta by Kite’s local partner Fosun in partnership with local insurer Chenxi Health, allowing up to 60% rebates on CAR-T costs if complete remission is not achieved.
Expert insight: “Cell and gene therapy market access in the Asia-Pacific region”
Watch the on-demand webinar
Supporting your Policy, Access, Value, and Evidence in the Asia-Pacific region
The expert APAC team at Avalere Health will help you bring out the best in your asset, using our extensive market access and disease area experience.
The APAC region is characterized by its dynamic nature and rapidly evolving markets. These are critical to the future growth plans of our biopharmaceutical clients and we are leading many groundbreaking market access projects in the region.
We’re based in Singapore, experienced, easy to talk to, and we love a challenge. Let us show you why our clients come back to work with us time and again. Contact us.
